Biogen To Report Q1 Earnings: Here's What To Expect
Photo by Viacheslav Bublyk on Unsplash
We expect Biogen (BIIB - Free Report) to beat expectations when it reports first-quarter 2023 results on Apr. 25 before the market opens. In the last reported quarter, the company delivered an earnings surprise of 15.38%.
The company’s earnings beat estimates in three of the last four quarters while missing expectations in one. The company delivered a four-quarter earnings surprise of 10.33%, on average.
Biogen’s stock has risen 31.8% in the past year against a decrease of 11.3% for the industry
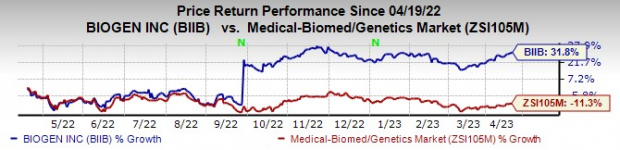
Image Source: Zacks Investment Research
Factors to Consider
Biogen’s sales in the first quarter are likely to have been hurt by lower sales of its multiple sclerosis drugs. Its multiple sclerosis revenues have been declining over the past few quarters. It is unlikely that MS sales have improved in the first quarter.
Among Biogen’s MS drugs, sales of Tecfidera are likely to have declined steeply, as multiple generic versions of the drug have been launched in the United States and new generic launches are ongoing in several EU countries.
Our estimates for first-quarter sales of Tecfidera are pegged at $234 million, while the Zacks Consensus Estimate is $269 million.
Sales of another MS drug, Tysabri are also likely to have declined in the first quarter due to increased competitive pressure and price reductions in certain European markets. A potential biosimilar of Tysabri may be launched in the United States and European markets as early as 2023.
Our estimates for first-quarter sales of Tysabri are $508.7 million, while the Zacks Consensus Estimate is pegged at $494 million.
Sales volumes of another MS drug Vumerity are expected to have been higher year over year in the first quarter driven by demand growth
Our estimates for first-quarter sales of Vumerity stand at $144.5 million, while the Zacks Consensus Estimate is pegged at $152 million.
Regarding supply constraints for Vumerity, Biogen said on the fourth-quarter conference call that it had resolved the previously reported manufacturing issues at its contract manufacturer and is also working on getting regulatory approvals for a secondary source of supply. Biogen does not expect a supply shortage in 2023. An update is expected on the first-quarter conference call.
Biogen receives royalties on U.S. sales of Roche’s (RHHBY - Free Report) MS drug, Ocrevus, which is also expected to have contributed to the top line.
Revenues from Biogen’s profit share of Roche’s drugs, Rituxan and Gazyva, are also likely to have declined in the quarter due to biosimilar competition for Rituxan.
Sales of Biogen’s spinal muscular atrophy drug, Spinraza improved in the fourth quarter as the company saw some signs of stabilization in inpatient use in the United States. Increased competition in the United States has been hurting demand and sales of Spinraza in the United States for the past few quarters. Outside the United States, volume growth in certain Asian markets is likely to have made up for increased competition in Europe. Our model estimates first-quarter sales of Spinraza to be $442.6 million while the Zacks Consensus Estimate is pegged at $428 million.
Biogen is likely to have recorded only minimal sales from the new Alzheimer’s drug, Aduhelm, in the fourth quarter. After the Centers for Medicare & Medicaid Services (“CMS”) denied all Medicare beneficiaries access to Aduhelm, in its final NCD decision, Biogen decided to substantially wind down commercial operations for Aduhelm, retaining only minimal resources to manage patients’ access programs.
Biosimilars revenues are expected to have been modest as the continued launch of Byooviz is expected to have made up for pricing pressure in certain markets. Our model estimates first-quarter sales of biosimilars to be $182.6 million, while the Zacks Consensus Estimate is pegged at $177 million.
Overall, the first quarter is a seasonally weaker quarter compared with the fourth quarter for the U.S. multiple sclerosis businesses due to channel dynamics and higher discounts and allowances. The fourth quarter of 2022 benefited from the favorable timing of some shipments of Spinraza, which is expected to have reversed in the first quarter of 2023. Operating expenses are also expected to have been higher.
Biogen is likely to have recorded only minimal sales from the new Alzheimer’s drug, Leqembi, an anti-amyloid beta protofibril antibody drug, for which the FDA had granted accelerated approval in January. The drug is not expected to contribute much to revenues until the CMS grants reimbursement for the drug under Medicare plans. This is expected to happen once Leqembi gets traditional approval. Eisai had submitted the sBLA to the FDA for traditional approval of Leqembi the same day it received the accelerated approval. In March, the FDA accepted the sBLA and granted priority review designation to the sBLA, with a decision expected on Jul 6, 2023.
Earnings Whispers
Our proven model does not conclusively predict an earnings beat for Biogen this time around. The combination of a positive Earnings ESP and a Zacks Rank #1 (Strong Buy), 2 (Buy), or 3 (Hold) increases the odds of an earnings beat. But that is not the case here.
Earnings ESP: Biogen’s Earnings ESP is -7.32% as the Zacks Consensus Estimate of $3.31 per share is higher than the Most Accurate Estimate of $3.07 per share. You can uncover the best stocks to buy or sell before they’re reported with our Earnings ESP Filter.
Zacks Rank: Biogen has a Zacks Rank #3.
More By This Author:
3 Lord Abbett Mutual Funds That Offer Appealing Returns
IBM Beats Q1 Earnings Estimates
Abbott's Q1 Earnings Beat Estimates, Margins Down
Disclosure: Zacks.com contains statements and statistics that have been obtained from sources believed to be reliable but are not guaranteed as to accuracy or completeness. References to any specific ...
more



