Brean Capital Weighs In On Medivation Following Update On Terrain Study
Medivation (NASDAQ: MDVN) is best known for developing therapies to treat serious diseases. Earlier in 2014 the biopharmaceutical company received FDA approval to expand the use of Xtandi, a prescription medicine used to treat men with prostate cancer.
At the European Association of Urology Congress on March 24, Medivation and Partner Astellas Pharmaceuticals (ALPMF) announced new data from its Phase 2 TERRAIN study, which evaluated Xtandi (enzalutamide) in comparison to Casodex (bicalutamide), a direct competitor of the treatment in metastatic prostate cancer. The company also announced final OS data from its Phase 3 PREVAIL trial in chemo-naïve mCRPC.
The TERRAIN study revealed that overall Prostate-specific antigen (PSA) progression was 13.6 months longer in patients being treated with Xtandi compared to 5.8 months for patients treated with Casodex. Patients being treated with Xtandi also showed a 50% PSA reduction over the course of 13 weeks, versus only a 21% reduction in patients being treated with Casodex in the same time-frame.
The PREVAIL trial treated male patients with chemotherapy-naive metastatic prostate cancer whose disease progressed despite having androgen deprivation therapy. After being treated with Xtandi, statistics showed the survival rate of these patients were overall beneficial.
Bertrand Tombal, M.D., Ph.D., chairman, stated, “This study demonstrates that starting patients on enzalutamide at the point when their castration-resistant prostate cancer becomes metastatic has the potential to prolong survival… The overall survival analysis from the PREVAIL trial confirms significant overall survival benefit despite many patients receiving subsequent treatments.”
On March 25, Brean Capital analyst Jonathan Aschoff weighed in on Medivation, reiterating a Buy rating on the stock and raising his price target from $125 to $142. He noted, “We remain bullish on Xtandi’s prospects to progress upstream in the prostate cancer treatment paradigm, and we look forward to Phase 2 STRIVE data sometime in the near future, which we view to be another significant clinical catalyst for Medivation. Given our optimism driven by consistently positive and differentiating data for Xtandi, we are raising our target price to $142 from $125.”
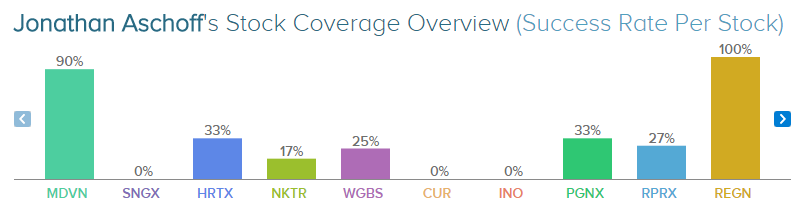
Aschoff has rated Medivation 10 times since November 2011, earnings a 90% success rate recommending the company and a +35.3% average return per recommendation. Overall, he has a 57% success rate recommending stocks and a +14.1% average return per recommendation.
Jonathan Aschoff is known for rating bio-pharmaceutical stocks, such as Regeneron (NASDAQ: REGN) and TG Therapeutics (NASDAQ: TGTX) . The analyst has rated Regeneron 10 times since April 2011, earning a 100% success rate recommending the company and a +23.3% average return per recommendation. Likewise, he has rated TG Therapeutics 8 times since May 2012, earning an 88% success rate recommending the stock and a +21.7% average return per recommendation.
Jonathan Aschoff sees a lot of potential in Medivation and Xtandi and is bullish on the stock. Do you agree with his latest recommendation based on his financial advice history?
On average, the top analyst consensus for Medivation on TipRanks is Strong Buy.
Disclosure: To see more recommendations for Netflix, visit more