Can-Fite Gearing-Up For Phase 2 NASH Program
TM editors' note: This article discusses a penny stock and/or microcap. Such stocks are easily manipulated; do your own careful due diligence.
CanFite Biopharma Ltd (CANF) is preparing to initiate a Phase 2 clinical study examining namodenoson (formerly CF102) for the treatment of NAFLD/NASH. Over the past several months, the company has been releasing preclinical data with namodenson demonstrating the drug reduces NAFLD Activity Score, micro- and macrovesicular fat deposits, decreased plasma ALT and triglycerides levels, prevents progression of liver fibrosis, and protects against ischemia and reperfusion injury in transplant models.
Quite simply, namodenoson looks like a pan-hepatic improver of liver pathology, applicable to earlier-stage liver diseases such as NAFLD and NASH, for the treatment of ischemia / reperfusion (I/R) injury in patients following partial hepatectomy, and in the treatment of primary liver cancer, including advanced hepatocellular carcinoma (HCC). I think this makes namodenoson an incredibly attractive asset for a potential larger biopharma company to acquired.
Below is a look at the current status of namodenoson and the preclinical data in NAFLD/NASH ahead of the planned Phase 2 trial later this year.
Quick Background on Namodenoson
Can-Fite's namodenoson (formerly CF102) is a selective A3 adenosine receptor (A3AR) agonist currently being investigated in as a second-line treatment for advanced hepatocellular carcinoma (HCC), the leading form of primary liver cancer, in subjects with Child-Pugh B cirrhosis. The 78-patient Phase 2 trial is currently recruiting patients in Israel and select countries in southeastern Europe. I'm expecting enrollment to complete in the next few months and for Can-Fite to report top-line data during the second half of 2017.
The anti-cancer effects of namodenoson are believed to stem from the activation of adenosine, mainly through binding to the A3 receptor (1). The mechanism of action mediated via A3AR agonism includes modulation of key signaling proteins, such as PI3K, GSK-3β, PKA, PKB/Akt, IKβ, and NF-кB, resulting in deregulation of the Wnt and the NF-кB pathways. In 2007, researchers at CanFite found that A3AR is expressed in very low levels in most normal tissues, but overexpressed in a number of different tumor types, including skin, colon, breast, renal, ovarian, and hepatic cancers (2). As such, targeting the A3AR through pharmaceutical intervention is a likely mechanism for apoptosis of cancer and inflammatory cells (3).
HCC is the lead indication for namodenoson. In fact, Can-Fite has received Fast Track designation for the drug as a second-line treatment for HCC, as well as been granted Orphan Drug status given the high unmet medical need and lack of effective treatment options for patients failing first-line therapy. HCC is a relatively small market in the U.S., with roughly 40,000 new cases each year (4). The opportunity in Europe looks similar; however, the American Cancer Society estimates more than 700,000 people are diagnosed with HCC cancer each year worldwide, with 450,000 or so in Asia.
Earlier in 2017, Can-Fite signed a distribution agreement with Chong Kun Dang Pharmaceuticals for namodenoson in South Korea. The deal came with a $0.5 million upfront payment and the potential to earn another $2.5 million in pre-commercial milestones. Can-Fite will also receive 23% royalty on namodenoson sales in South Korea from CKD. While this was not an enormous transaction, investors need to keep in mind that this was only for South Korea and that China is 27x the size. I believe there is a meaningful opportunity for Can-Fite to sign licensing and distribution agreements with namodenoson in areas like China and Southeast Asia that do get investors very excited.
Management has already out-licensed its other clinical-stage candidate, piclidenoson (formerly CF101), to Kwang Dong Pharmaceutical in Korea, so this management team has experience partnering with Asian pharmaceutical companies. Licensing deals that provide upfront cash to Can-Fite for Asia will help support the U.S. and EU development plans and allow the company to expand namodenoson into NASH, which is where I believe the true value of the asset lies.
Namodenoson For Liver Disease - An Exciting New Opportunity
Non-alcoholic steatohepatitis (NASH) is believed to be the next big global pandemic given the soaring rates of obesity, diabetes, and metabolic syndrome all over the world. NASH, also called "fatty liver", is a condition in which fat builds up in the liver causing inflammation. Prior to the presence of inflammation, the disease is simply referred to as non-alcoholic fatty liver disease (NAFLD), the most common form of liver disorder in the U.S.
The accumulation of macroglobular fat inside the liver causes oxidative stress that reduces the efficiency of the liver and can lead to increased liver enzymes such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST). Loss of liver efficiency and oxidative stress leads to inflammation, liver cell ballooning, and the development of NASH. Prolonged inflammation results in cirrhosis (scar tissue), liver failure, or liver cancer.
Rates of NAFLD and NASH are increasing in the U.S. in concert with increasing rates of obesity and diabetes. In fact, NASH is now the third leading cause of liver transplant in the U.S. According to the U.S. National Institutes of Health, an estimated 15-30% of Americans have fatty liver, with approximately 20% going on to develop NASH. The American College of Gastroenterology estimates 2-5% of adult Americans and up to 20% of those who are obese may suffer from NASH, putting the target population in the country between 5 and 10 million individuals. There are currently no U.S. FDA approved treatment options for NASH.
- Preclinical Data in NASH -
Preclinical data studying namodenoson in a mouse model of liver disease revealed the drug's capability to improve liver pathology in NAFLD and NASH. The data showed:
- Namodenoson had a statistically significant reduction in NAFLD Activity Score (NAS) compared to placebo. NAS was developed to provide a numerical score for patients who most likely have NASH. Accordingly, NAS is the sum of the separate scores for steatosis (0–3), hepatocellular ballooning (0–2), and lobular inflammation (0–3), with the majority of patients with NASH having a NAS score of ≥ 5. The namodenoson preclinical data shows statistical significance for both the high and low doses.
- Namodenoson reduced liver-to-body weight compared to placebo. Because of the excess accumulation of globular fat and inflammation of the liver, a NASH liver becomes enlarged and heavier than a normal healthy liver. A reduction in liver-to-body weight is evidence of reduced fat and inflammation in the liver and a sign that the drug is the improving disease condition.
- Liver sections from the placebo group exhibited severe micro- and macrovesicular fat deposits, hepatocellular ballooning and inflammatory cell infiltration, whereas the namodenoson treated group showed a significant decrease in steatosis, ballooning and lobular inflammation compared to the placebo group.
- Namodenoson decreased plasma ALT and triglycerides levels in the livers of NASH-model compared to placebo. This is a sign of improved liver function and efficiency. Separate studies with namodenoson demonstrated efficacy in the treatment of liver regeneration and function following liver surgery.
- Separate preclinical studies with namodenoson show the ability of the drug to inhibit, in a dose-dependent manner, the growth and proliferation of the liver fibrosis cells. This is particularly relevant to the clinical development of namodenoson targeting NAFLD/NASH.
- Representative photomicrographs of H&E-stained liver sections showed improved pathology in animals receiving namodenoson vs. placebo.
- Follow-up data released in February 2017 note that namodenoson prevents liver (hepatic) fibrosis progression in a mouse model of NASH disease. The namodenoson treated group exhibited normal liver under macroscopic view, no accumulation of fluid (ascites), a low fibrosis profile, and lower serum levels of transaminases as compared to the control group. In addition, liver protein extracts and mRNA for the alpha smooth muscle actin showed a significant anti-fibrotic effect in the namodenoson treated group as compared to the control group.
- Preclinical Data in Liver Ischemia / Reperfusion Injury -
In September 2016, Can-Fite announced additional preclinical data on namodenoson in liver disease was published in Molecular Medicine Report. The paper highlights the regenerative effects of namodenoson in a Wistar rat model of ischemia / reperfusion injury. The data show a higher regeneration rate in the namodenoson treatment group compared with the control group, suggesting that namodenoson had a positive effect on the proliferation of hepatocytes following hepatectomy (Ohana G, et al., 2016).
Ischemia / reperfusion (I/R) injury occurs when the cellular damage of a hypoxic organ is increased subsequent to the restoration of oxygen delivery. Warm I/R injury may arise clinically in hepatic surgery, liver transplantation, hypovolemic shock, and various types of toxic liver injury. These conditions are induced by liver hypoxia and may lead to ischemic hepatitis. Cellular hypoxia leads to the activation of the transcriptional regulatory, NF-кB, which triggers the release of other inflammatory mediators, including activated Kupffer cells and neutrophils, TNF‑α, IL‑1, and nitric oxide (NO) (5, 6). The mechanism of action of namodenoson, modulation of the Wnt and NF-кB signaling pathway through activation of A3AR, with downstream effects on PI3K, PKB/Akt, and IKβ reduces liver inflammation and may protect against hypoxia-related injury.
The figure below shows the serum concentrations of two liver enzymes, SGOT and SGPT, were significantly reduced in the namodenoson (CF102) treatment group compared to the control group 48 hours after surgery (P<0.05). This is evidence that CF102 helps prevent cellular injury and apoptosis following liver injury.

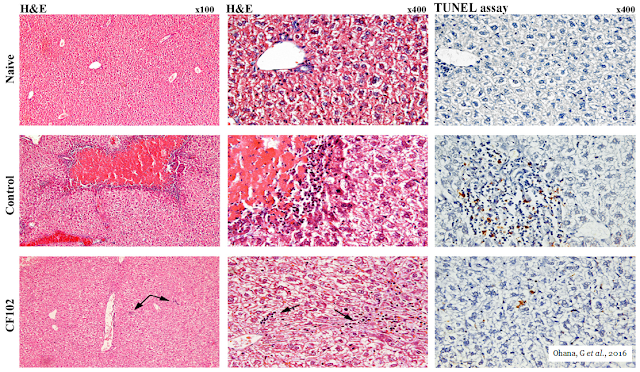
The picture below shows the histological evaluation of liver injury. No signs of necrosis, inflammation, or apoptosis were observed in the naïve liver. In the control group, large areas of necrosis and inflammatory cells were observed by H&E staining. The TUNEL assay revealed an abundance of cells undergoing apoptosis; however, in the namodenoson-treated group, no area of necrosis was observed, and only a few inflammatory cells appeared in the liver parenchyma (indicated by the arrows). Only a few cells were undergoing apoptosis in the TUNEL assay.
Additionally, hepatic regeneration rates were higher in the namodenoson treatment group compared with the saline‑treated control group 48 hours after the hepatectomy.

This preclinical data demonstrates that namodenoson when administered during liver reperfusion, may attenuate cellular injury, apoptosis, and the extent of necrosis in liver subjected to I/R. Additionally, namodenoson stimulated liver regeneration, which favored the survival of hepatocytes during I/R injury. Therefore, the ability of namodenoson to protect against hepatic I/R injury may include an anti-inflammatory and an anti-apoptotic effect, combined with an increased rate of liver regeneration.
Phase 2 Expected To Begin Later In 2017
CanFite plans to initiate a Phase 2 clinical program studying namodenoson in NAFLD/NASH in the next few months. The Phase 2 trial will be a multicenter, randomized, double-blinded, placebo-controlled, dose-finding study of the efficacy and safety of namodenoson in the treatment of NAFLD/NASH. Approximately 60 patients with NAFLD, with or without NASH, will be enrolled in three arms, including two different dosages of namodenoson and a placebo, given via oral tablets twice daily. The study's primary endpoints are likely to be percent change from baseline in liver triglyceride (fat) concentration measured by nuclear magnetic resonance spectroscopy (NMRS) and safety.
Secondary endpoints to be evaluated are the effects of namodenoson on metabolic abnormalities in subjects with NAFLD, including body weight, waist circumference, serum triglyceride and high-density lipoprotein cholesterol levels, and serum liver transaminase. In addition, an assessment of the pharmacokinetics (PK) of namodenoson and the A3 adenosine receptor (A3AR) biomarker will be evaluated prior to treatment and its correlation to patients' response to the drug will be analyzed upon study conclusion.
Furthermore, the exploratory objective of this study is to evaluate the effects of namodenoson on relevant biomarkers, such as adiponectin, leptin, C-reactive protein (CRP), and liver stiffness as determined by Fibroscan. The trial design is based on preclinical studies (discussed below) showing namodenoson's efficacy in reducing liver fat in NASH models as compared to placebo, improving liver function, and regenerating liver cells.
Conclusion
The mechanism of action for namodenoson looks like a pan-hepatic improver of liver pathology, applicable to earlier-stage liver diseases such as NAFLD and NASH, for the treatment of ischemia / reperfusion (I/R) injury in patients following partial hepatectomy, and in the treatment of primary liver cancer, including advanced hepatocellular carcinoma (HCC). Can-Fite anticipates initiating a Phase 2 trial in the next few months.
Big pharma has clearly shown a propensity to acquire early-stage assets for the treatment of NASH. In September 2016, Allergan paid $50 million to acquire Ankara because the potential exists that its preclinical asset, AKN-083, may have synergistic effects with cenicriviroc and evogliptin. Can-Fite's CF102 is orally administered, has a novel mechanism of action, and has been shown to be safe and generally well-tolerated in late-stage cancer patients.
Can-Fite's entire market capitalization is only $33 million and I have to believe that the majority of that value is Phase 3 piclidenoson for the treatment of rheumatoid arthritis and psoriasis. Namodenoson has the potential to dwarf piclidenoson if successfully developed for NASH. I think once Can-Fite initiates the Phase 2 study later this year, investors will start to view the story more and more as a "NASH play", and that should set the stage for a major revaluation higher.
Disclosure: Please see important information about BioNap, Inc. in our Disclaimer. BioNap owns stock ...
more


